Magnetic Nanoparticles for Next-Generation MRI Contrast Agents
1. Executive Summary
This study introduces a new class of amphoteric β-cyclodextrin-coated iron oxide magnetic nanoparticles (MNPs) optimized for biomedical imaging applications, particularly magnetic resonance imaging (MRI). The nanoparticles are synthesized using an innovative microwave–ultrasound-assisted protocol that enables precise control of size, morphology, and surface charge, resulting in uniform, stable, and cytocompatible nanostructures. By combining β-cyclodextrin and citric acid within a flexible amphoteric coating, the researchers achieved highly dispersible, hydrophilic nanoparticles capable of interacting effectively with biological environments. The coating preserves β-cyclodextrin’s inclusion capacity, enhancing molecular adsorption and potential drug-loading efficiency. Relaxometric analyses confirmed their superparamagnetic behavior and suitability as T2-weighted MRI contrast agents. The synthesis process, which integrates two enabling technologies—microwave heating and ultrasound irradiation—offers shorter reaction times, higher reproducibility, and improved nanostructural control compared with conventional methods. These results demonstrate the feasibility of scalable, biocompatible MNP production for advanced diagnostic imaging, combining efficiency, precision, and functional versatility for future medical and pharmaceutical applications.
2. Technology Description
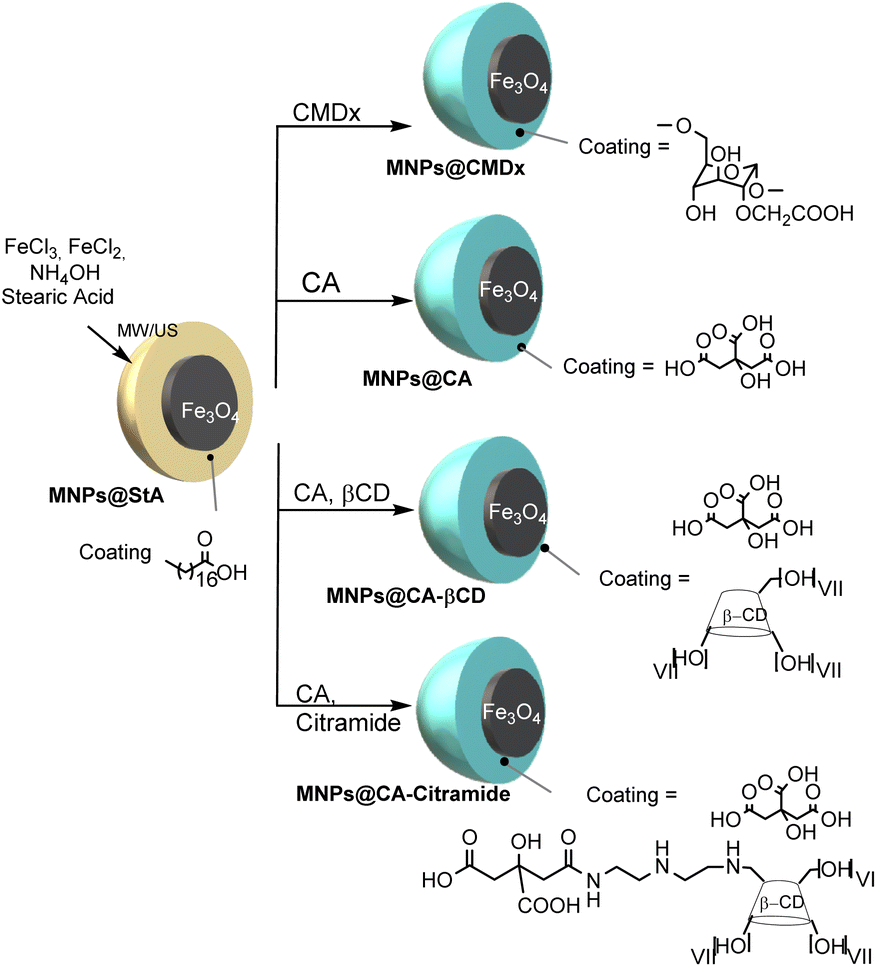
The developed technology consists of iron oxide magnetic nanoparticles coated with amphoteric β-cyclodextrin (βCD) and citric acid via a ligand-exchange process enhanced by microwave and ultrasound irradiation. This dual technique ensures homogeneous temperature distribution and effective particle dispersion, overcoming limitations typical of traditional co-precipitation synthesis. βCD functions as a host molecule forming inclusion complexes with organic compounds, while citric acid provides hydrophilicity and prevents aggregation through electrostatic repulsion. The resulting nanoparticles exhibit diameters below 90 nm, negative surface charges between −30 and −36 mV, and a superparamagnetic core characterized by Néel relaxation times within 10⁻¹⁰–10⁻⁷ s. Structural analyses using dynamic light scattering, thermogravimetric, and infrared spectroscopy confirmed coating integrity and uniform particle morphology. The microwave–ultrasound method reduces synthesis time from hours to minutes and enhances reproducibility and stability. The optimized βCD–citric acid–amine coating maintains βCD’s molecular inclusion ability, enabling simultaneous magnetic responsiveness and controlled molecular adsorption, thus integrating diagnostic and therapeutic capabilities within a single nanosystem.
3. Practical Applications
These nanoparticles are primarily designed for use as magnetic resonance imaging (MRI) contrast agents in medical diagnostics. Their hydrophilic and negatively charged surface ensures excellent colloidal stability in physiological media, extending circulation time and improving image contrast in T2-weighted imaging. The same structural and magnetic properties make them adaptable to other biomedical uses such as targeted drug delivery, cell tracking, magnetic hyperthermia, and tissue engineering. In drug delivery, β-cyclodextrin’s host–guest complexation can be used to encapsulate and release therapeutic molecules in response to environmental stimuli. Industrially, the microwave–ultrasound process offers an efficient, scalable synthesis pathway for pharmaceutical-grade nanomaterials with consistent performance. Beyond medicine, such nanoparticles could find applications in biosensing, environmental remediation, or magnetic separation processes where selective adsorption and controlled magnetization are required. Their multifunctionality and tunable properties position them as enabling materials at the intersection of imaging, therapy, and nanomanufacturing.
4. Key Benefits
The microwave–ultrasound synthesis protocol offers rapid, reproducible, and scalable production of uniformly coated nanoparticles, ensuring narrow size distribution and high magnetic homogeneity. The β-cyclodextrin–citric acid coating confers amphoteric properties, combining hydrophilicity, cytocompatibility, and inclusion capacity for organic molecules. Technically, these nanoparticles exhibit enhanced dispersibility and stability in aqueous and physiological conditions, maintaining negative surface charge that prevents aggregation. Their magnetic core provides high relaxivity, producing strong signal contrast in MRI while avoiding magnetic hysteresis, a hallmark of superparamagnetic systems. Economically, the method reduces energy consumption and process duration compared to conventional synthesis, facilitating industrial adoption. Operationally, it integrates diagnostic and potential therapeutic functions into a single nanosystem, reducing the need for multiple agents in biomedical workflows. The combination of controllable synthesis, multifunctional surface chemistry, and proven biocompatibility establishes a solid foundation for further preclinical and clinical development.
5. Challenges and Limitations
Despite its advantages, the technology still faces several constraints that may hinder immediate translation to industrial or clinical practice. The synthesis process, although efficient, requires precise control of microwave and ultrasound parameters to ensure reproducibility, and variations in frequency or power may lead to differences in nanoparticle morphology. The coating process demands rigorous optimization to balance stability, magnetic properties, and functionalization density without compromising inclusion capability. Regulatory approval for medical use requires extensive toxicological and pharmacokinetic studies, especially concerning long-term biocompatibility, clearance, and potential accumulation in organs. Scaling production under Good Manufacturing Practice (GMP) conditions may pose cost and equipment challenges. Additionally, while β-cyclodextrin coatings improve molecular adsorption, excessive surface modification could reduce magnetization efficiency or alter relaxation dynamics. Addressing these factors is essential for ensuring consistent quality, safety, and performance across batches, particularly for biomedical and pharmaceutical deployment.
6. Technology Readiness Level (TRL)
The described system is currently at TRL 4, corresponding to laboratory-scale validation of components in a controlled environment. Experimental synthesis, characterization, and preliminary in vitro testing have confirmed functionality, magnetic response, and cytocompatibility. The study includes comprehensive analyses of relaxometric profiles, magnetization behavior, and cellular viability using J774A.1 macrophage lines, demonstrating viability above 70% and strong T2 contrast in MRI phantoms. These findings validate the core technological concept and provide quantitative performance data. However, the transition to TRL 5 and beyond requires preclinical validation in animal models, reproducibility across scaled batches, and verification of long-term biocompatibility. Further optimization of ligand–exchange parameters and surface coatings under GMP-compatible conditions will be necessary to advance towards industrial pilot production and regulatory evaluation for diagnostic use.
7. Future Prospects
The research sets the foundation for a new generation of multifunctional magnetic nanomaterials for biomedical imaging and targeted therapy. Future developments will likely focus on refining the microwave–ultrasound synthesis parameters to achieve even narrower size distributions and scalable batch production. Advanced coatings could integrate specific targeting ligands, fluorescent markers, or therapeutic payloads, transforming these nanoparticles into true theranostic platforms that combine imaging, drug delivery, and hyperthermia capabilities. In the short term, optimization of β-cyclodextrin derivatives may further improve host–guest inclusion efficiency and molecular selectivity. Medium-term goals include preclinical animal testing to establish biodistribution and clearance pathways, while long-term objectives involve adaptation to multimodal imaging (MRI–PET, MRI–optical) and integration into personalized medicine frameworks. Beyond healthcare, similar synthesis protocols could be extended to environmental or industrial nanomaterials requiring controlled magnetism and biocompatibility.
8. Sources and Citations
Federica Calsolaro, Francesca Garello, Eleonora Cavallari, Giuliana Magnacca, Mikhail V. Trukhan, Maria Carmen Valsania, Giancarlo Cravotto, Enzo Terreno, Katia Martina, Amphoteric β-cyclodextrin coated iron oxide magnetic nanoparticles: new insights into synthesis and application in MRI, Nanoscale Advances, Royal Society of Chemistry, Issue 1, 2025, pp. 155–168. DOI: https://doi.org/10.1039/D4NA00692E.
9. Metadata & Tags
Title: Magnetic Nanoparticles for Next-Generation MRI Contrast Agents
Primary Technology Category: Biotechnology, Genomics & Human Enhancement
Secondary Technology Category: Health Technologies & Personalized Medicine
Date of Publication: 4 November 2024
Keywords: magnetic nanoparticles, β-cyclodextrin, microwave–ultrasound synthesis, MRI contrast, nanomedicine, amphoteric coatings, relaxivity, superparamagnetism, biocompatibility, nanotechnology
Primary Research Source: University of Turin (EuroBioImaging MultiModal Molecular Imaging Italian Node)
Year of Main Research: 2024
Technology Readiness Level (TRL): 4
Main Applications: Magnetic resonance imaging, drug delivery, and nanodiagnostics
Key Benefits: Highly stable, cytocompatible, and dispersible nanoparticles offering superior MRI contrast and scalable synthesis via enabling technologies
Target Audience: Pharmaceutical companies, medical device manufacturers, investors in diagnostic imaging technologies, public research institutions, and healthcare innovation agencies