Industrial Hydrogen Peroxide Without Metal Catalysts: A Carbon Electrode That Reaches Commercial Production Rates
Identifying the exact atomic defects in carbon that drive selective oxygen reduction — enabling metal-free hydrogen peroxide production at industrial scale
Executive Summary
Hydrogen peroxide — chemical formula H₂O₂ — is one of the most important commodity chemicals in the global economy. It is used to disinfect drinking water, bleach paper and textiles, clean silicon wafers in semiconductor manufacturing, sanitise food processing equipment, treat industrial wastewater, and serve as a precursor in pharmaceutical synthesis. Global production exceeds five million tonnes per year, supplying industries that together represent trillions of euros in annual economic activity. Despite its scale and importance, almost all hydrogen peroxide in the world is produced by a single industrial process developed in the 1930s — the anthraquinone oxidation process — which requires large centralised chemical plants, consumes hydrogen derived from natural gas, generates organic solvent waste that must be carefully managed, and produces a concentrated liquid so hazardous that its transport and storage are governed by international dangerous goods regulations.
This production model is deeply centralised by necessity rather than by choice. The chemistry is too complex and the product too dangerous to distribute easily to smaller users. Companies that need hydrogen peroxide — a municipal water treatment plant, a semiconductor fabrication facility, a textile mill, a pharmaceutical manufacturer — must purchase it from distant suppliers, manage the logistics of receiving hazardous shipments, and maintain on-site storage of a material that can corrode equipment, ignite organic materials, and release toxic oxygen if mishandled. The cost of this supply model is distributed across procurement budgets, facilities management, health and safety compliance, and insurance, and is rarely visible as a single line item but is substantial in aggregate.
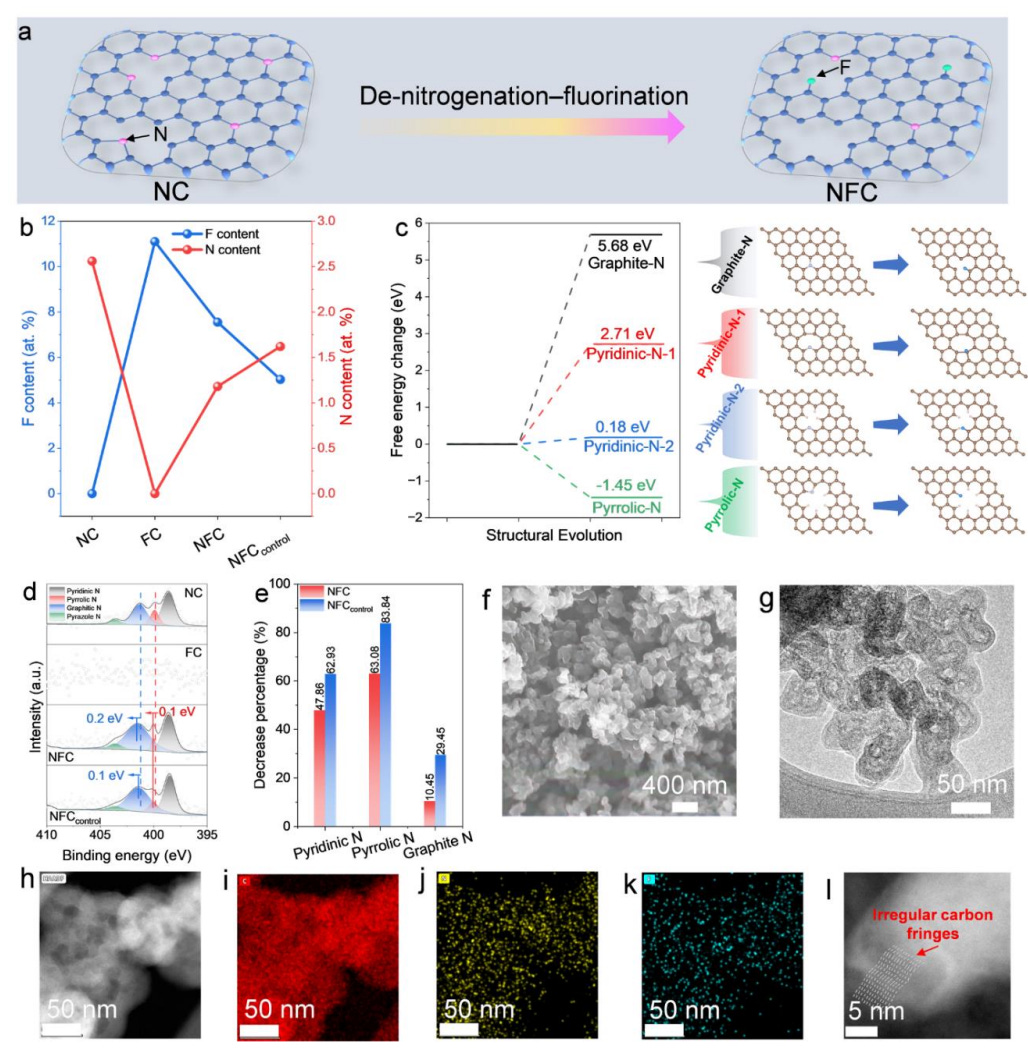
The research published in Nature Communications on 25 March 2026 by Yu, Bi, Yang and colleagues at the University of Central Florida offers a scientifically validated and commercially credible alternative to this centralised model. Their work demonstrates that hydrogen peroxide can be produced electrochemically — from oxygen in air and water, using electricity — at a carbon electrode that contains no metal whatsoever, and that the production rate is sufficient for industrial applications. This is not the first attempt at electrochemical hydrogen peroxide production, but it is the first to combine metal-free composition with industrial-scale current density through a rational, scientifically grounded design process rather than empirical trial and error. The scientific breakthrough is the identification of exactly which atomic configurations within the carbon material are responsible for the desired chemistry, which makes the catalyst designable rather than discoverable by chance. The practical consequence is a credible technology for on-site, renewable-powered hydrogen peroxide generation that could fundamentally change how companies in water treatment, semiconductors, food, and pharmaceuticals manage one of their most important chemical inputs.