How Artificial Intelligence Cuts Battery Development Time from Years to Weeks
A deep active learning framework with knowledge transfer triples lithium metal battery electrolyte lifespan while reducing the number of required experiments by more than 80%
Executive Summary
Developing a new electrolyte for a battery has traditionally required years of laboratory work. An electrolyte is the liquid or gel medium inside a battery through which charged atoms — ions — travel between the two electrodes during charging and discharging. The chemistry of electrolytes is extraordinarily complex. They must remain stable at high voltages, be chemically compatible with both electrodes simultaneously, and form a protective microscopic layer on the electrode surface — called the solid-electrolyte interphase — that prevents parasitic reactions from gradually destroying the battery during use. Finding the right combination of solvents, salts, and additives among the millions of possible candidate formulations has historically depended on the chemical intuition of experienced researchers, who test one candidate at a time and refine their hypotheses iteratively.
This process is slow, expensive, and inherently inefficient. A skilled electrolyte chemistry team typically needs three to five years to develop a new formulation suitable for a specific battery chemistry, and the cost of the experimental campaign — materials, characterisation, cell assembly, cycling tests — runs into millions of euros for each commercial-grade outcome. For lithium metal batteries, which represent the most promising route to dramatically higher energy density than current lithium-ion cells, the electrolyte challenge is particularly severe because lithium metal is extraordinarily reactive and the requirements on the electrolyte are correspondingly demanding.
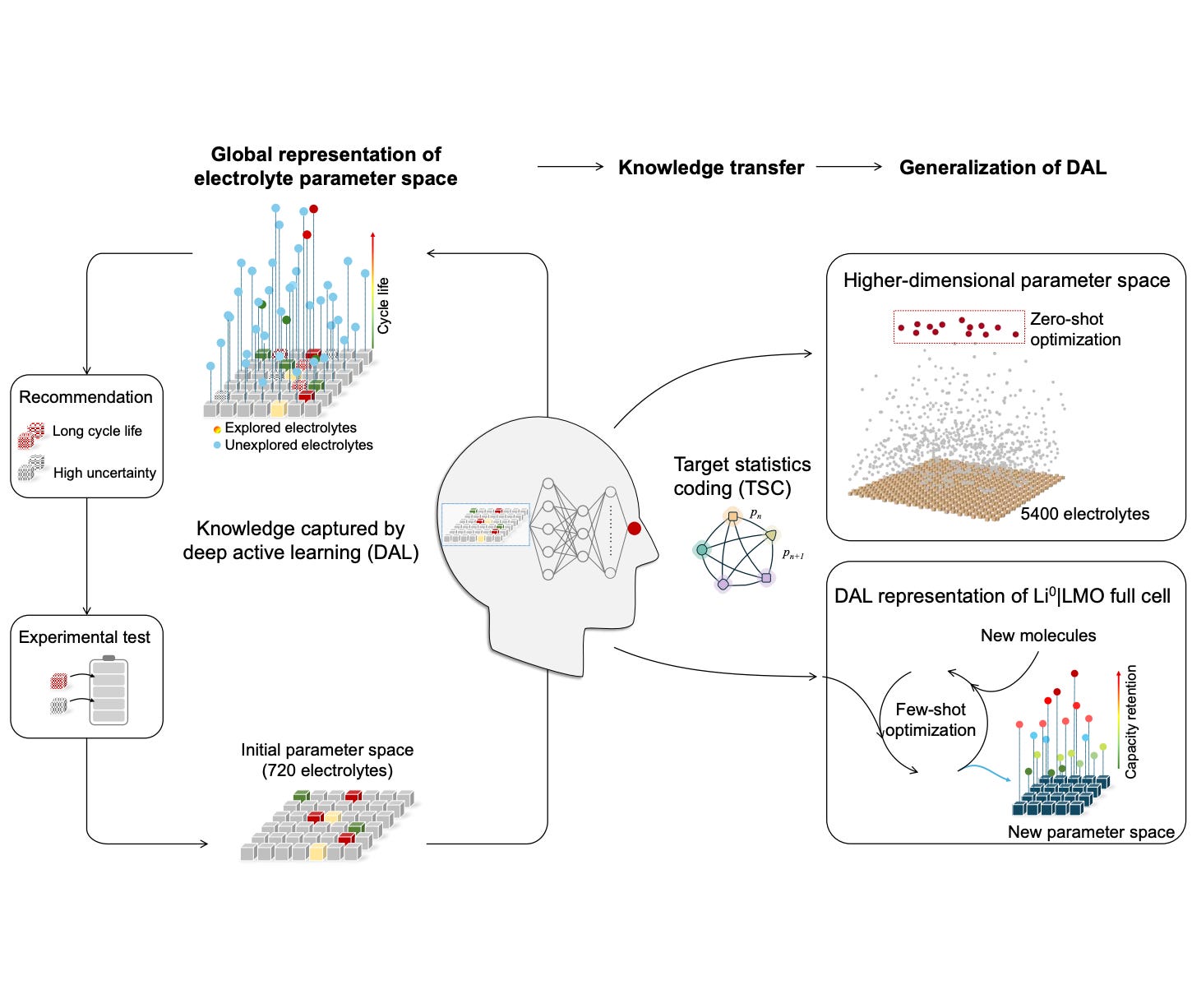
The research published in Nature Communications on 27 March 2026 by Hong, Wang, Harris, Jiang and colleagues from Tsinghua University and Lawrence Berkeley National Laboratory addresses this bottleneck directly. The authors present a machine learning framework that combines two complementary techniques to dramatically accelerate electrolyte discovery. The first is deep active learning, in which an artificial intelligence model iteratively selects the next experiment to run based on which formulation is most likely to provide new information about the underlying chemistry. The second is knowledge transfer, in which a model trained on one class of electrolyte formulations is adapted to a new class with minimal additional experimental data. The combined system identifies electrolyte formulations that triple the operational lifespan of lithium metal batteries while requiring more than eighty percent fewer experimental cycles than conventional empirical screening. For battery manufacturers, electrolyte material suppliers, and the investors funding them, this is not a future prospect — it is an immediately applicable methodology change in how electrolyte research is conducted.