Engineering Microbes to Work Together Without Falling Apart: A Control System That Stabilises Industrial Bacterial Cultures
A burden-aware multicellular RNA feedback control system stabilises engineered bacterial co-cultures for up to 24 hours, recovers 90% of growth rate after metabolic stress
Executive Summary
Industrial biotechnology produces a vast range of valuable products by cultivating engineered microorganisms — primarily bacteria and yeast — in large fermentation tanks. The list includes antibiotics, insulin, growth hormones, monoclonal antibodies, industrial enzymes used in detergents and food processing, amino acids used in animal feed, vitamins, biofuels, and an expanding range of bio-based chemicals and food ingredients. The global industrial biotechnology market exceeds three hundred billion euros annually and is growing faster than the chemical industry as a whole, driven by sustainability requirements, consumer demand for bio-based products, and pharmaceutical innovation.
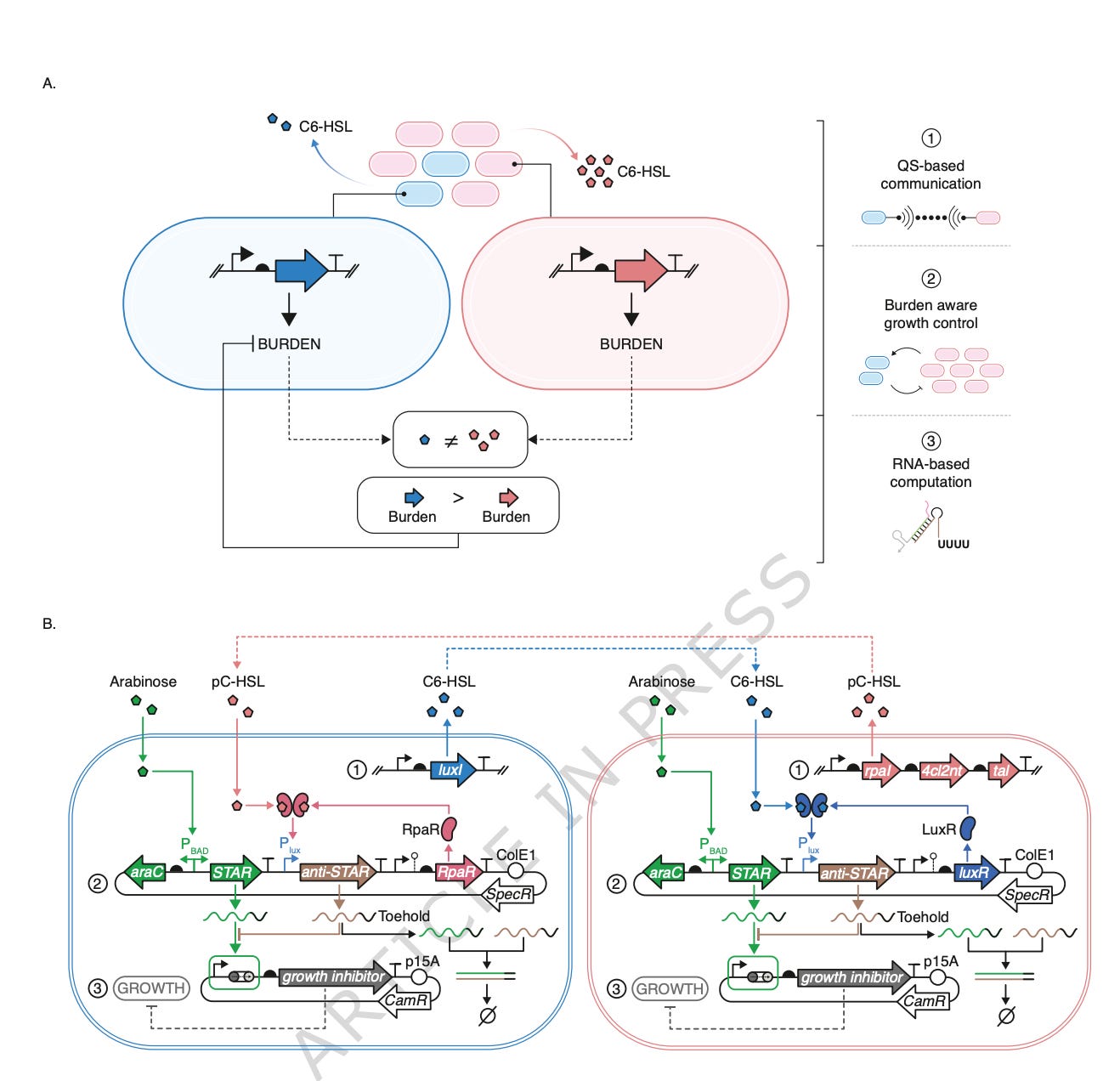
For decades, the standard approach has been to engineer a single bacterial strain to produce a single product. This works well for simple molecules, but it has fundamental limits when the desired product requires a complex biochemical pathway with many sequential enzymatic steps. Loading all of these steps onto one bacterial strain creates what microbiologists call a metabolic burden — the engineered cell must divert energy and resources from its own growth and survival to produce the introduced enzymes, and at some point the burden becomes so heavy that the cell stops growing or stops producing efficiently. The newer and more powerful approach is to use microbial consortia: multiple engineered strains working together, each carrying a portion of the biosynthetic pathway, dividing the metabolic labour and producing the final product collectively. In principle, this allows far more complex chemistries and dramatically higher yields than any single strain could achieve.
The persistent practical problem is that microbial consortia are unstable. In a fermentation tank, the different strains compete for the same nutrients and grow at different rates. The faster-growing strain inevitably crowds out the slower-growing one within hours, and the carefully designed division of labour collapses. The desired product yield drops, the process becomes unpredictable, and the consortium ceases to function as engineered. Stabilising the composition of a microbial consortium over the duration of an industrial fermentation has been one of the most important unsolved problems in synthetic biology for the past two decades, and its solution would unlock a substantial expansion of what industrial biotechnology can produce.
The research published in Nature Communications on 6 May 2026 by Boo, Mehta, Stan and colleagues at Imperial College London addresses this problem directly. The authors develop a multicellular feedback control system — a biological circuit distributed across multiple bacterial strains — that detects deviations from the target consortium composition and corrects them automatically, in real time, without any external monitoring or intervention. The system stabilises a two-strain Escherichia coli co-culture over the full duration of a twenty-four-hour batch fermentation, recovers up to ninety percent of growth rate after the introduction of a heavy metabolic load, and increases protein production yield in the slower-growing strain by up to eighty-one percent. For industrial biotechnology companies, this is a foundational engineering capability that converts microbial consortia from a laboratory curiosity into a deployable production platform.